“Forensic” analyses in fish markets


“Forensic” analyses in fish markets



Molecular “Forensic” analyses in Peruvian fish markets reveal that multiple dolphin species are marketed despite legal measures banning cetacean takes
(Please, refer to the original publication at the bottom of this page for references & details)
Summary

Fig. 1. Dolphin meat sold in local Peruvian fish markets. This meat is a substantial source of revenues for the local communities but over-exploitation of dolphin stocks may jeopardize the survival of the dolphin populations. For by-catch reduction to be sustainable, wide involvement of fishers and efficient communication about management objectives and methodology are of paramount importance. Unfortunately, the most effective methods have so far been detrimental to the local fishery industry. This not only affects the fishermen’s livelihoods, but also exacerbates the divide and decreases trust between scientists and managers on one hand, and fishermen and local communities on the other. In nations, such as Peru, where small cetacean by-catches may reach population-threatening levels, a wide debate among the public, the stakeholders, and the experts about by-catch mitigation measures is greatly desirable.
The problem of ‘by-catch’ in fishery activities
Since the development some 60 years ago of cheaper and stronger (nylon) fishing gear, fishing became much less selective: a staggering amount of non-target marine life is hauled up every day with the catch, and then usually discarded overboard dead or dying. Occasionally, these animals, known as ‘by-catch’ or ‘incidental catch’ are subsequently and opportunistically sold, but might ultimately become new targets of fishery activities. Over 300,000 small whales, dolphins, and porpoises die from entanglement in fishing gears each year, making by-catch the single largest cause of mortality for small cetaceans and pushing several species to the verge of extinction. Drift and set gillnets pose the greatest threat to cetaceans due to their relatively low cost and ubiquitous use both in coastal and offshore waters.
Monitoring and quantifying by-catches might help increase the pressure on involved parties to establish less destructive fishing practices. While attention has tended to focus on the control of legal and illegal international trade as a threat to biodiversity, much of the world’s commerce in wildlife and fisheries is domestic and largely unregulated. Small-scale fisheries, for example, employ over 80% of the world’s 38 million fishers and provide about half of all fish caught for human consumption. These fisheries occur primarily in developing nations, and their documentation and management are locally limited or non-existent, precluding evaluation of their impact on non-targeted fauna. In such cases, surveys of large commercial markets, the end-point of the supply chain, can be an effective means of estimating true levels of exploitation or takes. Maximizing the efficiency of such actions may involve the use of molecular genetic methods (when animals are not landed openly) for estimating the actual number of animals killed as well as for gathering basic parameters such as the species, the sex, and possibly the population of origin of each sampled individual (see HERE our previous molecular genetic characterization of dusky dolphin and Burmeister’s porpoise populations).
Cetaceans ‘by-catch’ in Peru
By-catches and directed takes of small cetaceans in Peru have been monitored at regular intervals since 1985. Early studies clearly pointed to increasing numbers of captured animal. In 1990, a ministerial decree outlawed the exploitation of small cetaceans, but this rule was widely ignored and total annual catches reached an estimated 15,000–20,000 specimens in the period 1990–1993. The Peruvian Ministry of Fisheries issued a stricter decree in 1994, that was accompanied by greater efforts to publicize and enforce the measures; as a result, overt landings ceased and cetacean meat gradually disappeared from view at public markets, although unknown quantities of meat were commercialized illegally. Finally, a 1996 national law prohibited entirely all captures and trade for five marine odontocetes -- Lagenorhynchus obscurus (Dusky dolphin), Phocoena spinipinnis (Burmeister’s porpoise), Tursiops truncatus (Bottlenose dolphin), Delphinus capensis (Long-Beaked Common Dolphin), and D. delphis (Short-beaked common dolphin) -- and two riverine dolphins (Sotalia fluviatilis, the ‘Tucuxi’, and Inia geoffrensis, the Amazon river dolphin). Although these measures along with public campaigns, are likely to have undermined incentives for directed takes, they also seem to have pushed fishers to adopt new illegal practices. Once the new protective regulations were enforced, by-caught specimens were either disposed at sea, butchered on board to bait longlines and gillnets that target large sharks and rays, or covertly landed as filleted meat for human consumption. Simultaneously, the use of cetaceans as bait has been a rapidly expanding phenomenon given the high prices and low availability of traditional bait fish and the continuing supply of cetacean carcasses. Ironically, national legislation hardly influenced incidental captures, given that no adjustments in fishing techniques were established to mitigate net entanglements, but it made it far more difficult to record such events. Accordingly, the daily number of cetaceans that could be examined in local markets has drastically declined over the last 20 years. On the other hand, an on-board survey in 2005–2007 showed that small cetaceans are still captured in high numbers.
Molecular genetic analyses of meat samples from Peruvian fish markets
In our new study, we used molecular genetic approaches to analyse 182 samples of meat and skin of unidentified small cetaceans collected at Peruvian markets between July 2006 and April 2007. We used molecular genetic analyses (DNA sequencing and genotyping of microsatellite markers) to determine (i) the relative species composition of the sampled animals, (ii) if individuals were sampled more than once, and (iii) the sex of each sampled animal. Our results are integrated in the table below (2006-2007 (market)) together with statistics from present and past onboard and market surveys. Absolute number of animals (and percentage) are indicated.
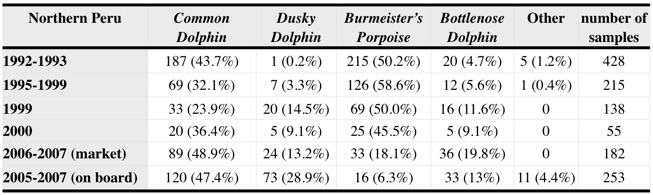
These analyses indicate that the same species continue to be caught and marketed. In addition, a large decrease over time of the relative abundance of Burmeister’s porpoises was detected. Fluctuations can be due to shifts in species distribution, changes in preferred fishing grounds (inshore vs. offshore habitat, lower vs. higher latitudes), or to population depletion.
Our analyses also (i) indicate evidence of gene flow, or retention of ancestral haplotypes, among the different D. delphis populations around the world in consistence with previous studies, and (ii) confirm our previous molecular genetic analyses (more information available HERE): Peruvian waters host populations of both Dusky Dolphins and Burmeister’s porpoises that are each genetically isolated from con-specific southern hemisphere populations, thus warranting special conservation and management status. Finally, our present analyses indicate (in accordance with previous morphological, ecological, behavioral, and distributional evidence) the existence of a wide-ranging Bottlenose Dolphin offshore population off Peru and Chile, distinct from smaller inshore populations.
Based on 7 microsatellite loci and the fact that there are no means to conserve meat at the markets, we identified 8 individuals that have been sampled more than once. This is very different from the situation in Korea where the half-life of whale meat is 1.8 months. This kind of information is critical, because it indicates that the optimal frequency with which markets should be monitored can greatly vary depending of the local storage practice, in addition to differences in muscle mass of the cetaceans involved (25–30 kg/dolphin compared to several tons/whale).
Sex bias
The molecular assays for sexing individuals indicate a highly skewed sex ratio for all species (about 80:20% males:females). However, and contrary to the species assignment assays, these results should be taken with caution because pseudo-autosomal chromosomic regions have not been fully characterized in cetaceans. Note that recent analyses of samples from minke whales (Balaenoptera acutorostrata) harvested by the Japanese and Korean fleets also suggested a male-biased exploitation at certain areas. A similar result was obtained from a 17 years long survey on stranded bottlenose dolphins (Tursiops truncatus) in the Mississippi sound region, as well as for the short fin mako sharks in the southeast Pacific Ocean, resulting from yet unexplained sexual segregation mechanisms. The phenomenon has been observed in several other cetacean species (see our original publication for references).
Conclusions
In Peru, as in many other countries, legislation banning all takes, including by-catch, in fishing activities has ironically hidden conservation problems and made population management more challenging because monitoring becomes highly problematic when fishers continue catching cetaceans but utilize or dispose of carcasses clandestinely. Thus, analysis of interview data or official reports may lead to the erroneous conclusion that by-catch is rare or has largely been phased-out, as has been claimed for Peru. It is likely that sample collection at local markets as well as on-board observations are the only means to obtain reliable estimates of the extent of the phenomenon. However, on-board observer programs are expensive and can be difficult to implement and maintain. So far, outside North America, Western Europe, Australia, and New Zealand, there have been very few observer programs designed to monitor cetacean by-catch. Such programs would involve high costs for developing countries. Nevertheless, the by-catch produced in these areas should not be ignored.
Our results from molecular analyses of samples collected in three Peruvian markets (and compared to existing data from past surveys, as well as to an on-board monitoring program that was conducted in parallel) indicate that penalizing small-cetacean indirect takes in Peru has not eliminated the problem, but rather concealed it. A similar effect from equivalent regulatory measures has been observed for other marine species, like sea turtles and seabirds, suggesting that legislative or other regulatory protective measures often have limited efficiency for reducing the impact of fisheries on marine fauna.
For by-catch reduction to be sustainable and operational, wide involvement of fishers and efficient communication about management objectives and methodology are of paramount importance. For different fisheries, circumstances and regions, numerous ways of reducing by-catch have been suggested, from modifications of fishing gear, use of active acoustic alarms or ‘pingers’, seasonal closures of specific fishery activities to complete closure of fisheries (UN General Assembly 1991). Unfortunately, the most effective methods have so far been detrimental to the local fishery industry. This not only affects the fishermen’s livelihoods, it also exacerbates the divide and decreases trust between scientists and managers on one hand, and fishermen and local communities on the other. Furthermore, measures to reduce by-catches of one species can endanger others: for example, switching from gillnets to longline fleets could reduce the impact on small cetaceans, but would probably put local turtle populations at risk. In nations, such as Peru, where by-catch levels of small cetaceans may reach population-threatening levels, a wide debate among the public, the stakeholders, and the experts about by-catch mitigation measures is greatly desirable.
Full Original Publication
Please, consult the full publication for references and much additional information
✓Tzika A.C., D’Amico E., Alfaro-Shigueto J., Mangel J.C., Van Waerebeek K. & M.C. Milinkovitch
Molecular identification of small cetacean samples from Peruvian fish markets
Conservation Genetics 11: 2207-2218 (2010)
In the media (in French)
✓Un petit dauphin du Pérou victime de la surpêche
Par Anton Vos,
✓Une étude génétique quantifie le braconnage des dauphins
Other related publications
✓Mangel JC, Alfaro-Shigueto J, Van Waerebeek K et al.
Small cetacean captures in Peruvian artisanal fisheries: high despite protective legislation
Biological Conservation 143: 136–143 (2010)
✓Cassens I., Van Waerebeek K., Best P. B., Crespo E.A., Reyes J. & M. C. Milinkovitch
The phylogeography of dusky dolphins (Lagenorhynchus obscurus): a critical examination of network methods and rooting procedures
Molecular Ecology, 12: 1781-1792 (2003)
✓Cassens I., Van Waerebeek K., Best P.B., Tzika A., Van Helden A.L., Crespo E.A. & M. C. Milinkovitch
Evidence for male dispersal along the coasts but no migration in pelagic waters in dusky dolphins (Lagenorhynchus obscurus)
Molecular Ecology, 14 : 107-121 (2005)
✓Cassens I., Mardulyn P. & M. C. Milinkovitch
Evaluating Intraspecific “Network” Construction Methods Using Simulated Sequence Data: Do Existing Algorithms Outperform the Global Maximum Parsimony Approach?
Systematic Biology 54: 363-372 (2005)
✓Rosa S., Milinkovitch M.C., Van Waerebeek K., Berck J., Oporto J., Alfaro-Shigueto J., Van Bressem M.F., Goodall N. & I. Cassens
Population structure of nuclear and mitochondrial DNA variation among South American Burmeister’s porpoises (Phocoena spinipinnis)
Conservation Genetics 6: 431–443 (2005)
✓Mardulyn P., Cassens I. & M. C. Milinkovitch
A comparison of methods for constructing evolutionary networks from intraspecific DNA sequence
Pages 104-120 (Chapter 5) in ‘Population Genetics for Animal Conservation’ (Bertorelle G, Bruford M.W.,
Hauffe H.C., Rizzoli A., & Vernesi C., eds.),
Cambridge University Press 2009

Back to the
“SoutH-American dolphins” page
Back to the
“Conservation Genetics” page
Back to the
“LANE”
introduction page


FACTS
Species:
‣Lagenorhynchus obscurus
‣Phocoena spinipinnis
‣Tursiops truncatus
‣Delphinus delphis
‣Delphinus capensis
Locations:
‣Peru

PUBLICATIONS
‣Conservation Genetics 11: 2207-2218 (2010)

PEOPLE INVOLVED
FROM MICHEL
MILINKOVITCH’S LAB
‣Athanasia Tzika

MAIN COLLABORATORS
Koen Van Waerebeek
Peruvian Centre for Cetacean Research (CEPEC), Museo de Delfines, Pucusana, Lima, Peru
Julio Reyes
Areas Costeras y Recursos Marinos (ACOREMA), Pisco, Peru
Joanna Alfaro-Shigueto & Jeffrey Mangel
Asociacion Pro Delphinus, Lima, Peru




